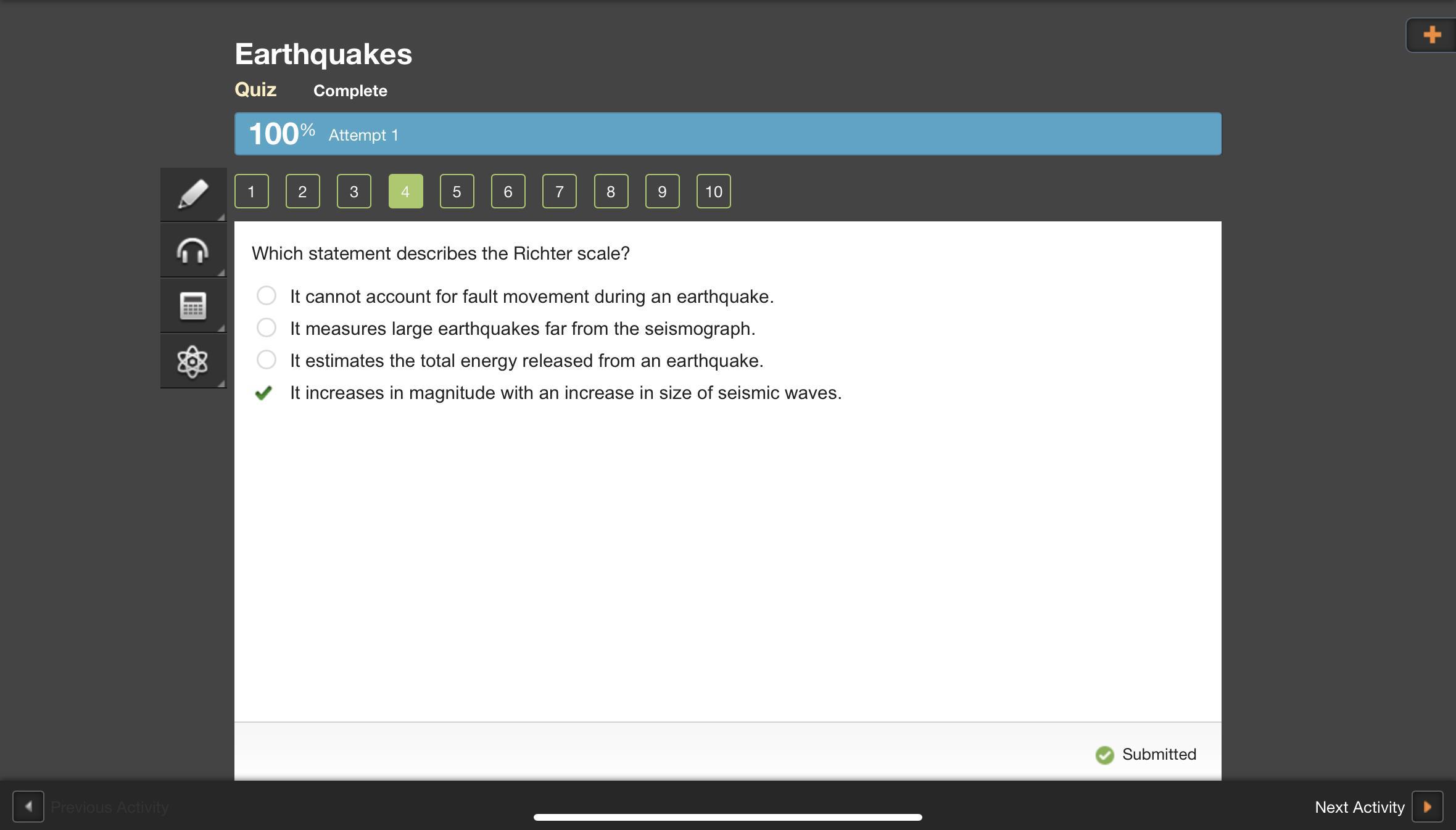
Which statement describes the Richter scale?
It cannot account for fault movement during an earthquake.
It measures large earthquakes far from the seismograph.
It estimates the total energy released from an earthquake.
It increases in magnitude as amount of damage increases.
Answers
Answer:
It measures large earthquakes far from the seismograph.
Explanation:
It uses a seismograph to measure earthquakes far from the seismograph where seismic waves are used to determine the magnitude of an earthquake.
Answer: d. It increases in magnitude with an increase in size of seismic waves.
Explanation:
Related Questions
The type of stem cells that are thought to be pluripotent are found in
A.) embryos
B.) developed tissues
C.) cord blood
D.) the placenta.
Answers
Answer:
Embryonic stem cells
Explanation:
Answer:
C.)
Explanation:
( Hope this helps 0_0 )
if 300.0g of chlorine is reacted how much calcium hydroxide is needed to react with it
Answers
Answer:
334.7g of Calcium Hydroxide are needed
Explanation:
Based on the reaction:
2Ca(OH)2 + 2Cl2 → Ca(ClO)2 + CaCl2 + 2H2O
2 moles of chlorine react with 2 moles of calcium hydroxide.
To solve this question we must find the moles of chlorine in 300.0g. These moles = Moles of Ca(OH)2. With the moles of calcium hydroxide we can find its mass as follows:
Moles Cl₂ (Molar mass: 70.90g/mol)
300.0g Cl₂ * (1mol / 70.90g) = 4.2313 moles Cl₂ = Moles Ca(OH)₂
Mass Ca(OH)₂ (Molar mass: 74.093g/mol)
4.2313 moles Ca(OH)₂ * (74.093g / mol) =
334.7g of Calcium Hydroxide are needed5. A sample of an unknown metal has a mass of 120.4 g. As the sample cools from 90.5°C to 25.7°C, it releases 7020. J of energy. What is the specific heat of the sample? Identify the metal among those in the data table below. Finally, is this endothermic or exothermic?
Answers
Answer: The specific heat capacity of the sample is [tex]0.899 J/g^{o}C[/tex] and as heat is released in this reaction so it is exothermic in nature.
Explanation:
Given: Mass = 120.4 g
Heat energy released = -7020 J
Initial temperature = [tex]90.5^{o}C[/tex]
Final temperature = [tex]25.7^{o}C[/tex]
Formula used is as follows.
[tex]q = m \times C \times (T_{2} - T_{1})[/tex]
where,
q = heat energy
m = mass of substance
C = specific heat capacity
[tex]T_{1}[/tex] = initial temperature
[tex]T_{2}[/tex] = final temperature
Substitute the values into above formula as follows.
[tex]q = m \times C \times (T_{2} - T_{1})\\-7020 J = 120.4 g \times C \times (90.5 - 25.7)^{o}C\\C = \frac{-7020 J}{120.4 \times (-64.8^{o}C)}\\= \frac{7020 J}{7801.92} J/g^{o}C\\= 0.899 J/g^{o}C[/tex]
When heat is released in a process or reaction then it means it is exothermic in nature.
Thus, we can conclude that the specific heat capacity of the sample is [tex]0.899 J/g^{o}C[/tex] and as heat is released in this reaction so it is exothermic in nature.
Is Zinc sulfide ionic or covalent?
Answers
Hikers have noticed that a sealed bag of potato chips puffs up when taken to the top of a mountain. Suppose that at the valley floor below, the air pressure is 1.0 atm, temperature is 25°C, and the volume of the chip bag is 0.985 L. At the top of the mountain, the temperature is 22°C and the chip bag has puffed up to 1.030 L.
What is the air pressure on top of the mountain?
__ atm
Answers
Answer:
0.95
Explanation:
trust
3. a. How many moles of NaCl are dissolved in 152 mL of a solution if the concentration of the solution is 0.364 M?
Answers
Answer:
Good luck man
Explanation:
Good luck man
2.3g of lithium chloride in 500cm3 of solution
Answers
Answer:
0.109M
Explanation:
Find molarity of the solution:
Molarity is defined as the ratio between the moles of the solute (Lithium Chloride in this case) per liter of solution. To solve this question we have to find the moles of LiCl and its volume as follows:
Moles LiCl -Molar mass: 42.394g/mol-
2.3g * (1mol / 42.394g) = 0.0543 moles
Volume in liters:
500cm³ * (1L / 1000cm³) = 0.500L
Molarity:
0.0543 moles / 0.500L =
0.109M
Explain what happens when there is a lot of extra greenhouse gases in our atmosphere. Explain what happens when there's not enough greenhouse gases in our atmosphere. Based on those answers, are greenhouse gases inherently good or bad? Explain.
Answers
Explanation:
Here is why: If the atmosphere contains too much of these gases, the whole Earth becomes a hotter and hotter greenhouse. The atmosphere holds onto too much of the heat at night instead of letting it escape into space. Then, the next day, the Sun heats Earth's surface even more.
A substance has a boiling point of 78 °C. Which of the following is true about the substance? (5 points) a It will also change from a gas to a solid at 78 °C while the gas loses energy. b It will also change from a gas to a solid at 78 °C while the gas gains energy. c It will also change from a gas to a liquid at 78 °C while the gas loses energy. d It will also change from a gas to a liquid at 78 °C while the gas gains energy.
Answers
Answer: If a substance has a boiling point of [tex]78^{o}C[/tex] then it is true that it will also change from a gas to a liquid at 78 °C while the gas loses energy.
Explanation:
The temperature at which vapor pressure of a liquid substance becomes equal to the atmospheric pressure is called boiling point of substance.
At the boiling point, liquid phase and vapor phase remains in equilibrium.
This means that as liquid phase changes into vapor phase and also vapor phase changes into liquid phase at the boiling point.
Thus, we can conclude that if a substance has a boiling point of [tex]78^{o}C[/tex] then it is true that it will also change from a gas to a liquid at 78 °C while the gas loses energy.
is zinc an example of a element or compound?
Answers
Answer:
Zinc is an element.
Explanation:
Compounds are composed of two or more elements
Answer:
element
Explanation:
zinc is an element with atomic number 30
10OIS
Save for Later
1
Yusef is doing an experiment to find out how the amount of sunlight affects the growth of a seedling. He has three identical pots
containing grass seeds he planted at the same time. He puts them in windows that receive different amounts of sunlight each day.
Every day. Yusef looks at the pots and records his observations. What is the independent variable in Yusef's experiment?
OA type of soil
OB type of seedling
Ос.
amount of water
OD
amount of sunlight
Reset
Next Question
Answers
Answer:
asdffafAFSAfdsdf
Explanation:
Which of these elements will successfully create a 1:1 bond with Boron (B)
Phosphorus (P)
Helium (He)
Nitrogen (N)
Aluminum (AI)
Answers
Answer:
Option D, Aluminum
Explanation:
Valency of boron and hence it requires three electron to complete its outermost shell.
Valency of Aluminum is also three and it can donate three electrons to boron to complete the outermost shell of itself as well as the boron.
Hence, Aluminum and Boron will form 1:1 bond with each other. (Al-B)
Hence, option D is correct
A student uses visible spectrophotometry to determine the concentration of CoCl2(aq) in a sample solution. First the student prepares a set of CoCl2(aq) solutions of known concentration. Then the student uses a spectrophotometer to determine the absorbance of each of the standard solutions at a wavelength of 510nm and constructs a standard curve. Finally, the student determines the absorbance of the sample of unknown concentration. The original solution used to make the solutions for the standard curve was prepared by dissolving 2.60g of CoCl2 (molar mass 130.g/mol) in enough water to make 100.mL of solution. What is the molar concentration of the solution
Answers
Answer:
0.200 M
Explanation:
Step 1: Given data
Mass of CoCl₂ (m): 2.60 gMolar mass of CoCl₂ (M): 130. g/molVolume of solution (V): 100. mL (0.100 L)Step 2: Calculate the moles (n) of CoCl₂ (solute)
We will use the following expression.
n = m / M
n = 2.60 g / (130. g/mol) = 0.0200 mol
Step 3: Calculate the molar concentration of the solution
Molarity is equal to the moles of solute divided by the liters of solution.
M = n/V
M = 0.0200 mol/0.100 L = 0.200 M
What are the 3 most valuable metals in the United States? A. Lead b. Gold c. Iron d.Copper e.Coal f.Silver g.Titanium
Answers
Answer:
Gold, Silver and Titanium
Explanation:
The most valuable metals in the list are:
Gold:
Its value makes it a good investment option. Gold can be used in many different ways, like in jewelry, electronics, medicine, etc.
Silver:
Similar to the case of gold, besides the common value of silver in jewelry, it can be also used in other ways, because it has a large thermal and electrical conductivity.
Finally, the titanium: It's price is actually close to the copper one, but for the cost of work with titanium (Titanium has a really large melting point, then melting it into ingots needs a lot of energy), the price of titanium is increased.
I can't do Miscellaneous calculations!
E.g.
How many moles of oxygen atoms are present in 2.5 mole of water molecules?
Answers
take the unknown (the one that the qn is asking for) and ratio it with the known variable
and then put in the mol that you know for the known variable and just do math to get the answer for the unknown
Please heeelp to solve these problems
Answers
Answer:
Q.89
Alkane - CnH(2n+2)
given that 8 H = > 8= 2n+2
therefore n= 3
C3H8 = 12×3 + 8×1= 36 +8 = 44
what are the effect of water pollition
Answers
a railroad car with a mass of 90,000 kg is traveling along a straight, horizontal track at a constant speed of 2 m/s. the car collides with a second railroad car, also with a of 90,000 kg that is initially at rest. The railroad cars stick together after the collision, as shown in the figure.
1) 0.5 m/s
2) 1 m/s
3) 2 m/s
Answers
Answer:
I think it's 2.
Explanation:
The weight should transfer half of it's velocity to the other one. or its 1
Describe one way in which advances in space technology have improved our understanding of objects in space.
Answers
Answer:
Technology helps us in many ways.
Explanation:
We can now research the world even more effectively and at a higher level of sophistication thanks to technological advancements. Telescopes, on the other hand, enable us to peer into the far corners of the universe and detect very large objects such as stars. Electron microscope enable us to see smaller objects such as cells and organisms.
How does competition among different species lead to extinction?
Answers
[tex]\huge{\textbf{\textsf{{\color{pink}{An}}{\red{sw}}{\orange{er}} {\color{yellow}{:}}}}}[/tex]
Interspecific competition often leads to extinction. The species that is less well adapted may get fewer of the resources that both species need. As a result, members of that species are less likely to survive, and the species may go extinct.
Thanks Hope it helps.what does molecule mean?
a.) A pure substance, only one type of atom
b.) Two or more atoms held together by chemical bonds
c.)Composed of two or more separate elements
d.)The smallest part of matter still retaining chemical properties
Answers
A pure substance, only one type of atom
Answer:
(a) a pure substance, only one type of atom
Can u guys help me I have an f in science and this will help me a lot with my grade.
Answers
Answer:
First slide:
1) diffusion
2)water
3)high
4)low
5)balance
Second slide:
1)movement
2)without
3)energy
4)high
5)low
6)equilibrium
Final slide:
1)passive
2)energy
3)randomly
4)high
5)low
6)equilibruim
7)permeable
8)Osmosis
9)water
Hopes this helps! Good luck!
I will give the brainliest
The concentration of a saturated solution of calcium carbonate, CaCO3, is 8.7 × 10-9 Molar. What is the Ksp of calcium carbonate?
A.
4.7 × 10-13
B.
9.3 × 10-5
C.
7.6 × 10-17
D.
8.7 × 10-17
Answers
Answer:
C
Explanation:
x = 8.70 x 10^-9 M
Ksp = [x][x] = x² = (8.70 x 10^-9)^2 = 7.57 x 10^-17
4
If a gas takes up 250 ml at 800. torrand 50.°C, how much space will it take up at 400 torr and 25°?
A 125 ml
B 500 ml
0.46 L
D 0.542 L
Answers
Answer: The space occupied by the gas at 400 torr and [tex]25^{o}C[/tex] is 250 mL.
Explanation:
Given: [tex]V_{1}[/tex] = 250 mL, [tex]P_{1}[/tex] = 800 torr, [tex]T_{1} = 50^{o}C[/tex]
[tex]V_{2} = ?[/tex], [tex]P_{2}[/tex] = 400 torr, [tex]T_{2} = 25^{o}C[/tex]
Formula used is as follows.
[tex]\frac{P_{1}V_{1}}{T_{1}} = \frac{P_{2}V_{2}}{T_{2}}[/tex]
Substitute the values into above formula as follows.
[tex]\frac{P_{1}V_{1}}{T_{1}} = \frac{P_{2}V_{2}}{T_{2}}\\\frac{800 torr \times 250 mL}{50^{o}C} = \frac{400 torr \times V_{2}}{25^{o}C}\\V_{2} = 250 mL[/tex]
Thus, we can conclude that space occupied by the gas at 400 torr and [tex]25^{o}C[/tex] is 250 mL.
Whch is more acidic, a solution with a pH of 2 or 5
Answers
Answer:
A solution with a pH of 2
Explanation:
The lower a pH is in a liquid, the more acidic it is. 7 is the neutral pH for a liquid. We usually drink around that pH for water. A pH higher than 7 expresses that the liquid is a base.
Answer:
A solution with a pH of 2 is more acidic.
Explanation:
The solution with a pH of 2 is more acidic because on the pH scale as you get closer to zero, the more acidic the solution becomes.
Why do you think volcanoes are found in a line ?
Answers
Answer:
i never seen a volcano before
during prophase a group of fibers called what forms the cell
Answers
Answer:
During prophase, the nucleus disappears, spindle fibers form, and DNA condenses into chromosomes ( sister chromatids ). During metaphase, the sister chromatids align along the equator of the cell by attaching their centromeres to the spindle fibers.
Explanation:
If I beat it, I ain't wearin' a johnny (Hah)
Adeola wanna roll with a geezer (With a geez)
Is it me or the lifestyle, sweetheart?
Actually, I don't give a shi (Nah)
I'm a rapper now, might as well live in it (Live in it)
Write out a “for every” statement that relates the amount of methane to the amount of oxygen gas (CH4 + 2O2 = CO2 + 2H2O)
Answers
Answer:
For every one molecule of methane, one molecule of oxygen is required to release one molecule of CO2 and 2 molecule of H2O
Explanation:
As we can in the balanced equation below -
CH4 + 2O2 = CO2 + 2H2O
One molecule of CH4 reacts with two molecule of oxygen to release one molecule of CO2 and 2 molecule of H2O.
Hence, it can be said
For every one molecule of methane, one molecule of oxygen is required to release one molecule of CO2 and 2 molecule of H2O
What is the molality of a solution in which 0.32 moles aluminum chloride has been dissolved in 2,200 g water
Answers
A man needs to lift a heavy box on the back of a truck or push up a ramp or lift it straight up from the ground to use around he won't have to move the box over a longer distance what did is the benefit of using a ramp to get the box